 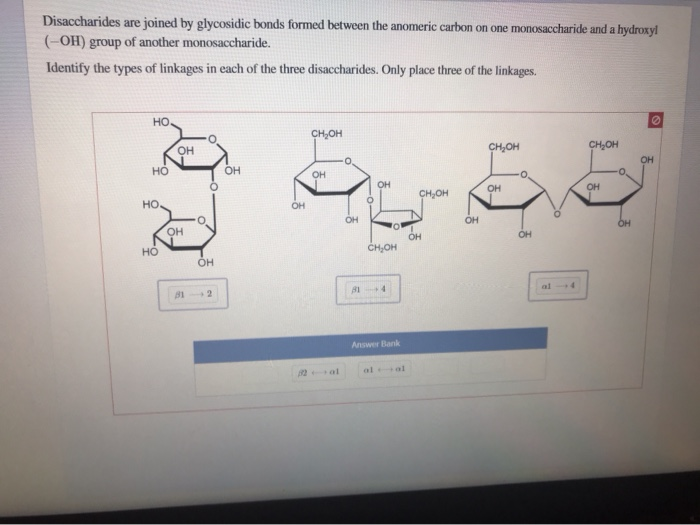 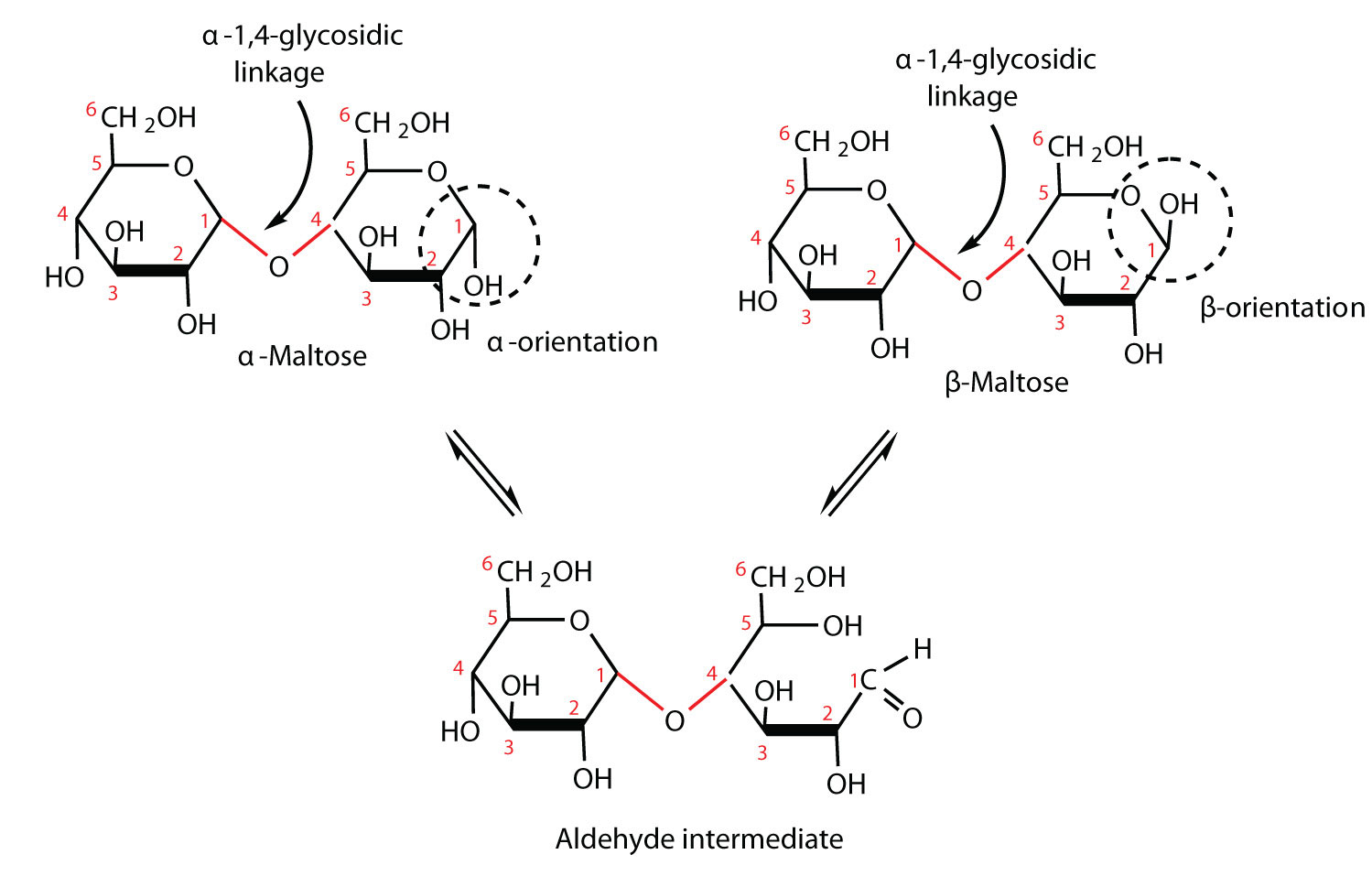
In the alpha form, the hydroxyl group on the anomeric carbon and the CH 2OH group on the last carbon within the ring are located on opposite sides of the ring, and in the beta form, they are located on the same side.Ī similar naming convention is used for bonds linking the monosaccharides to other molecules.īonds formed below the ring structure are known as alpha linkages, whereas those above the ring are known as beta linkages. Anomers are structural isomers that differ in the configuration at the carbonyl carbon. These carbon-oxygen ring structures can exist as two anomers: alpha and beta. The carbon attached to the reactive aldehyde or ketone is also known as the anomeric carbon. 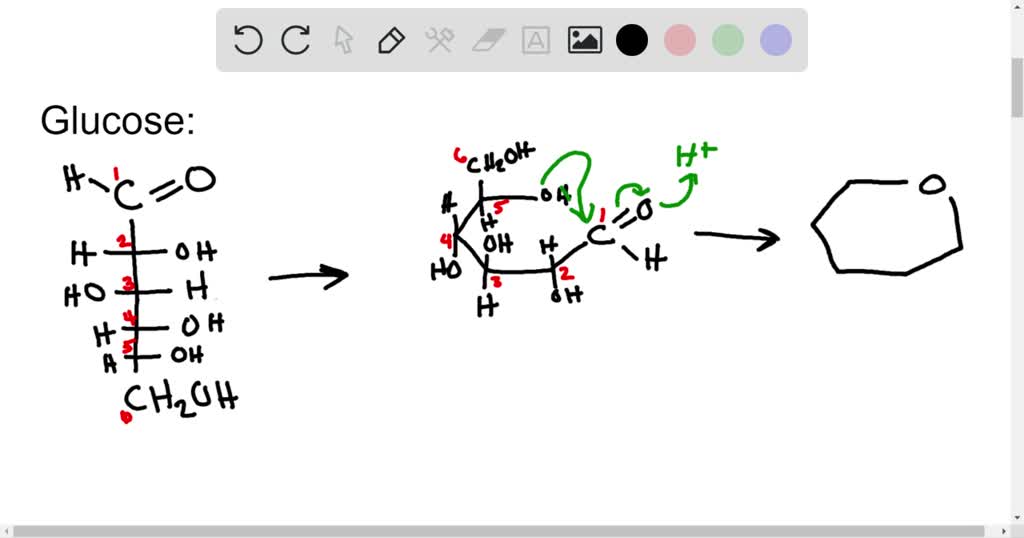
Ring structures form when the carbonyl group reacts with a hydroxyl group at the opposite end of the molecule through a condensation reaction. Sugars can form non-planar ring structures and in nature mostly exist in this cyclic form. Each of the carbons can be numbered starting with the number one from the end closest to the carbonyl carbon. Monosaccharides usually contain three to seven carbon atoms in their chains. Those having an aldehyde group are called aldoses and those having a ketone group are called ketoses. Complex carbohydrates are polymers called polysaccharides.Ī monosaccharide can be classified as an aldose or a ketose by its functional group. Simple carbohydrates are monomers called monosaccharides and dimers called disaccharides. These molecules get their name from the empirical formula of many monosaccharides that have two atoms of hydrogen and one atom of oxygen for every carbon. Carbohydrates are compounds composed of carbon, hydrogen, and oxygen. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
